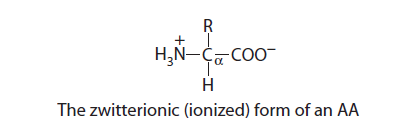20 Amino Acids and Their Functions
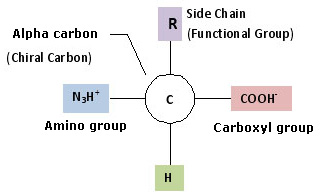
All 20 standard amino acids share the same core structure — an amino group, a carboxyl group, and a hydrogen atom bonded to a central carbon. What makes each one unique is the fourth attachment: the R group, or side chain. Below are all 20, organized by side chain chemistry, with their principal roles in the body.
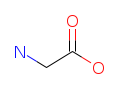 | Glycine (G/Gly). The simplest amino acid and the only one without a chiral center. Despite its small size, glycine is remarkably versatile: it makes up about 35% of collagen by mass, acts as an inhibitory neurotransmitter in the brainstem and spinal cord, and was famously detected in the tail of comet Wild 2 by NASA's Stardust mission. Read more about Glycine. |
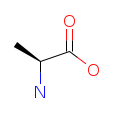 | Alanine (A/Ala). One of the body's primary glucogenic amino acids and a key energy source for muscle tissue. Alanine plays a central role in the glucose-alanine cycle, shuttling nitrogen from working muscles to the liver for safe disposal, while returning glucose to fuel further activity. Read more about Alanine. |
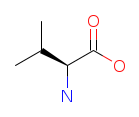 | Valine (V/Val). One of the three branched-chain amino acids (BCAAs), essential for muscle metabolism, tissue repair, and nitrogen balance. Valine is metabolized directly in muscle tissue rather than the liver, making it a rapid fuel source during physical exertion. Read more about Valine. |
 | Leucine (L/Leu). The most potent activator of muscle protein synthesis among the branched-chain amino acids — it activates the mTOR pathway through upstream sensing mechanisms, signaling muscle cells to initiate protein synthesis. Also involved in wound healing, bone repair, and growth hormone signaling. Read more about Leucine. |
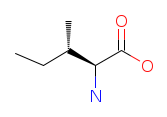 | Isoleucine (I/Ile). Essential for hemoglobin synthesis and the regulation of blood sugar and energy levels. Like leucine and valine, isoleucine is metabolized primarily in muscle, and it plays an important role in immune function and recovery from physical stress. Read more about Isoleucine. |
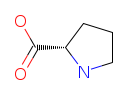 | Proline (P/Pro). A structurally unique amino acid: its side chain loops back to form a ring with the backbone nitrogen, creating a rigid kink that disrupts alpha-helices and forces sharp turns in protein chains. Proline is a critical component of collagen and cartilage, supporting joints, tendons, and ligaments. Read more about Proline. |
 | Methionine (M/Met). Carries a unique distinction: protein synthesis in all domains of life is initiated with methionine — or its modified form, formylmethionine, in bacteria and organelles — though the initiator methionine is frequently removed post-translationally. An antioxidant with lipotropic properties relevant to hepatic lipid metabolism, and the primary donor of methyl groups in dozens of biochemical reactions, including the synthesis of creatine, carnitine, and epinephrine. Read more about Methionine. |
 | Phenylalanine (F/Phe). An essential precursor to tyrosine and, through it, to dopamine, norepinephrine, and adrenaline. Critical for healthy nervous system function. The D-form of phenylalanine has been investigated for its potential role in pain modulation — one of the few cases where a D-amino acid has pharmacological relevance in humans. Read more about Phenylalanine. |
 | Tyrosine (Y/Tyr). The direct precursor to dopamine, norepinephrine, adrenaline, and thyroid hormones. The relationship between tyrosine availability and dopaminergic function has been studied in the context of cognitive performance under physiological stress; some studies report associations with working memory and alertness in high-demand conditions. Read more about Tyrosine. |
 | Tryptophan (W/Trp). The sole dietary precursor to serotonin and, downstream, melatonin — its availability is a rate-limiting factor in serotonin biosynthesis, linking intake to serotonergic and circadian function. Among the least abundant of the 20 standard amino acids in most dietary proteins, it also has the lowest recommended daily intake of all essential amino acids. Read more about Tryptophan. |
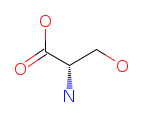 | Serine (S/Ser). A versatile amino acid with roles in both structure and metabolism. Serine is a key component of phospholipids — the molecules that form cell membranes — and is found in myelin sheaths, the insulating layer around nerve fibers. It also participates in the synthesis of purines, pyrimidines, and other amino acids. Read more about Serine. |
 | Threonine (T/Thr). Essential for the synthesis of collagen and elastin. Threonine has lipotropic properties associated with hepatic lipid metabolism, and plays a role in immune function through its involvement in immunoglobulin synthesis. It was the last of the 20 standard amino acids to be identified, isolated in 1935. Read more about Threonine. |
 | Cysteine (C/Cys). Forms the disulfide bonds that lock proteins into their three-dimensional shapes — the molecular staples of protein architecture. A potent antioxidant and detoxifier, cysteine (and its supplement form N-acetylcysteine) can bind heavy metals and neutralize free radicals. It is also the amino acid that makes permanent hair curls chemically possible. Read more about Cysteine. |
 | Asparagine (N/Asn). The first amino acid ever isolated — extracted from asparagus juice by French chemists Vauquelin and Robiquet in 1806. Asparagine plays a key role in protein glycosylation (the attachment of sugar chains to proteins) and serves as a nitrogen transport molecule between tissues. Read more about Asparagine. |
 | Glutamine (Q/Gln). The most abundant free amino acid in human blood. Glutamine is the primary fuel for intestinal cells and fast-dividing immune cells, making it critical for gut integrity and immune defense. It also serves as a major nitrogen shuttle between organs and supports muscle protein preservation during periods of stress or illness. Read more about Glutamine. |
 | Lysine (K/Lys). Essential for collagen cross-linking and a major component of muscle protein. Lysine is also the precursor to L-carnitine, the molecule responsible for transporting fatty acids into mitochondria for energy production — making it important for both cardiovascular health and fat metabolism. Read more about Lysine. |
 | Arginine (R/Arg). A key intermediate in the urea cycle, converting toxic ammonia into urea for safe excretion. Arginine is also the only biological precursor to nitric oxide — the signaling molecule that mediates vasodilation, blood pressure regulation, and vascular function. Its role in nitric oxide biosynthesis has made it a subject of research interest in exercise physiology. Read more about Arginine. |
 | Histidine (H/His). Found in unusually high concentrations in hemoglobin, where it plays a direct role in oxygen binding and release. Also a precursor to histamine — the signaling molecule involved in immune response, gastric acid secretion, and neurotransmission in the brain. Read more about Histidine. |
 | Aspartate (D/Asp). A central player in both the urea cycle and the citric acid cycle, connecting amino acid metabolism to energy production. Aspartate acts as an excitatory neurotransmitter and donates nitrogen atoms in the biosynthesis of purines and pyrimidines — the building blocks of DNA and RNA. Read more about Aspartate. |
 | Glutamate (E/Glu). The principal excitatory neurotransmitter in the vertebrate nervous system, central to synaptic transmission throughout the brain and spinal cord. Also responsible for the distinctive savory umami taste — its sodium salt, MSG, was isolated from kombu seaweed in 1908 by Kikunae Ikeda. Read more about Glutamate. |